Class 12 Physics Complete Guide – All Chapters with Formulas & Examples
Master Class 12 Physics (CBSE 2025–26) with clear explanations, key formulas, derivations, and solved examples — Electrostatics, Magnetism, Optics, Modern Physics and more.
Overview: What You Will Learn in Class 12 Physics
Class 12 Physics is one of the most important subjects you will study in your school career. It forms the foundation for engineering, medicine, research, and technology—and its concepts appear in every major competitive exam, from JEE and NEET to university entrance tests worldwide. The CBSE Class 12 Physics syllabus (2025–26) is divided into 9 units covering 14 chapters, worth 70 marks in theory and 30 marks in practicals.
This guide walks through every chapter in a way that actually makes sense—starting with the concept, building the mathematics, and giving you the key formulas, typical exam questions, and tricks that will help you score full marks. Whether you're studying for the first time or revising the night before an exam, this is your complete reference.
| Unit | Chapters | Theory Marks |
|---|---|---|
| Unit I – Electrostatics | Ch 1: Electric Charges & Fields | Ch 2: Electrostatic Potential & Capacitance | 16 |
| Unit II – Current Electricity | Ch 3: Current Electricity | 17 |
| Unit III – Magnetic Effects & Magnetism | Ch 4: Moving Charges & Magnetism | Ch 5: Magnetism & Matter | 17 |
| Unit IV – EM Induction & AC | Ch 6: Electromagnetic Induction | Ch 7: Alternating Current | — |
| Unit V – EM Waves | Ch 8: Electromagnetic Waves | 18 |
| Unit VI – Optics | Ch 9: Ray Optics | Ch 10: Wave Optics | — |
| Unit VII – Dual Nature of Radiation | Ch 11: Dual Nature of Radiation & Matter | 12 |
| Unit VIII – Atoms & Nuclei | Ch 12: Atoms | Ch 13: Nuclei | — |
| Unit IX – Electronic Devices | Ch 14: Semiconductor Devices | 7 |
Unit I: Electrostatics — Electric Charges, Fields & Potential
Electrostatics is the study of charges at rest. Everything starts with a simple observation: rub a plastic ruler on your hair and it attracts tiny bits of paper. That invisible force is the electric force, and it is one of the four fundamental forces of nature. Unit I (Chapters 1 & 2) carries 16 marks—the highest single unit—so understanding it deeply pays off enormously.
Chapter 1 – Electric Charges and Fields
All matter is made of atoms containing protons (+) and electrons (−). When a body has unequal numbers of protons and electrons it becomes charged. The SI unit of charge is the Coulomb (C). The fundamental charge (charge on one proton or electron) is $e = 1.6 \times 10^{-19}$ C. Charge is always quantised ($q = ne$, where $n$ is an integer) and conserved (total charge of an isolated system never changes).
Coulomb's Law
The force between two point charges $q_1$ and $q_2$ separated by distance $r$ in a medium of permittivity $\varepsilon$ is:
where $k = 9 \times 10^9$ N m² C⁻² and $\varepsilon_0 = 8.85 \times 10^{-12}$ C² N⁻¹ m⁻² is the permittivity of free space. The force is along the line joining the charges: attractive for unlike charges, repulsive for like charges. For a system of multiple charges, use the superposition principle: the total force on any charge is the vector sum of forces due to all other individual charges.
Electric Field
The electric field $\vec{E}$ at a point is the force per unit positive test charge placed at that point:
SI unit: N C⁻¹ (or V m⁻¹). Electric field lines start on positive charges and end on negative charges; they never cross each other. For a dipole (two equal and opposite charges $±q$ separated by distance $2a$), the field on the axial line is $E_{axial} = \frac{2kp}{r^3}$ and on the equatorial line is $E_{equatorial} = \frac{kp}{r^3}$, where $p = q \cdot 2a$ is the dipole moment.

Gauss's Law
Gauss's Law is a powerful tool for calculating electric fields of highly symmetric charge distributions. It states that the total electric flux through any closed surface (Gaussian surface) equals the enclosed charge divided by $\varepsilon_0$:
| Charge Distribution | Gaussian Surface | Electric Field |
|---|---|---|
| Infinite line charge (λ C/m) | Coaxial cylinder of radius r | $E = \dfrac{\lambda}{2\pi\varepsilon_0 r}$ |
| Infinite plane sheet (σ C/m²) | Pillbox/cylinder | $E = \dfrac{\sigma}{2\varepsilon_0}$ |
| Thin spherical shell (Q) | Concentric sphere (outside, r > R) | $E = \dfrac{Q}{4\pi\varepsilon_0 r^2}$ |
| Thin spherical shell (Q) | Concentric sphere (inside, r < R) | $E = 0$ |
Chapter 2 – Electrostatic Potential and Capacitance
The electric potential $V$ at a point is the work done per unit positive charge in bringing a test charge from infinity to that point:
SI unit: Volt (V = J C⁻¹). Potential is a scalar quantity. The potential difference $V_{AB} = V_A - V_B$ is the work done per unit charge in moving from $B$ to $A$. The relationship between field and potential is $E = -\frac{dV}{dr}$—the field points from high to low potential.
A capacitor stores electric charge. The capacitance $C$ is defined as $C = Q/V$, measured in Farads (F). For a parallel-plate capacitor with plate area $A$, separation $d$, and dielectric constant $K$:
Energy stored in a capacitor: $U = \frac{1}{2}CV^2 = \frac{Q^2}{2C} = \frac{1}{2}QV$. When capacitors are combined in series: $\frac{1}{C_s} = \frac{1}{C_1} + \frac{1}{C_2} + \cdots$ and in parallel: $C_p = C_1 + C_2 + \cdots$
Unit II: Current Electricity — Circuits, Resistance & Kirchhoff's Laws
When charges move in an ordered fashion through a conductor, we have electric current. Current electricity (Chapter 3) explains how circuits work, why some materials resist current flow more than others, and the rules that let us analyse any circuit no matter how complex. This unit carries significant exam weight and produces a large number of numerical problems.
Electric Current and Drift Velocity
Electric current is the rate of flow of charge: $I = \frac{dQ}{dt}$, measured in Amperes (A). In metallic conductors, current flows due to the drift of free electrons opposite to the applied electric field. The drift velocity $v_d$ is tiny—typically $\sim 10^{-4}$ m/s—yet the electric signal propagates at nearly the speed of light because the field is established throughout the wire almost instantaneously.
where $n$ is the number density of free electrons, $A$ is the cross-sectional area, $e$ is electron charge, and $v_d$ is drift velocity.
Ohm's Law and Resistance
For ohmic conductors, the current through a conductor is directly proportional to the potential difference across it at constant temperature: $V = IR$. The resistance $R$ depends on geometry and material:
where $\rho$ (resistivity) is a material property, $L$ is length, and $A$ is cross-sectional area. Resistivity varies with temperature: $\rho_T = \rho_0 [1 + \alpha(T - T_0)]$, where $\alpha$ is the temperature coefficient of resistance. For metals, $\alpha > 0$ (resistance increases with temperature); for semiconductors and insulators, $\alpha < 0$.

Kirchhoff's Laws
Kirchhoff's laws are essential for analysing multi-loop circuits:
- KCL (Junction Rule): The algebraic sum of all currents at a junction is zero: $\sum I = 0$. This is a statement of conservation of charge—charge cannot accumulate at a node.
- KVL (Loop Rule): The algebraic sum of potential differences around any closed loop is zero: $\sum V = 0$. This reflects conservation of energy—a charge returning to its starting point has the same potential energy.
Key Circuit Elements & Formulas
| Concept | Formula | Notes |
|---|---|---|
| Resistors in Series | $R_s = R_1 + R_2 + R_3$ | Same current through each; voltages add |
| Resistors in Parallel | $\dfrac{1}{R_p} = \dfrac{1}{R_1} + \dfrac{1}{R_2}$ | Same voltage across each; currents add |
| EMF & Terminal Voltage | $V = \varepsilon - Ir$ | r = internal resistance of cell |
| Power Dissipated | $P = VI = I^2 R = V^2/R$ | Unit: Watt (W) |
| Wheatstone Bridge (balanced) | $\dfrac{P}{Q} = \dfrac{R}{S}$ | No current through galvanometer |
| Metre Bridge (slide wire) | $\dfrac{R}{S} = \dfrac{\ell}{100 - \ell}$ | ℓ = balance length from left |
Unit III: Magnetic Effects of Current and Magnetism
Moving charges create magnetic fields, and magnetic fields exert forces on moving charges. This elegant symmetry—charge creates field, field acts on charge—underpins the entire technology of electric motors, generators, transformers, and MRI machines. Unit III (Chapters 4 & 5) is high-weightage (17 marks combined with Unit IV) and rich in derivations.
Chapter 4 – Moving Charges and Magnetism
The Lorentz Force is the total force on a charged particle in combined electric and magnetic fields:
The magnetic force $\vec{F}_B = q\vec{v} \times \vec{B}$ is always perpendicular to both $\vec{v}$ and $\vec{B}$—it does no work and cannot change the particle's speed, only its direction. A charged particle in a uniform magnetic field moves in a circle (or helix if it has a component along $\vec{B}$). The radius of this circular path is:
Biot–Savart Law
The Biot–Savart Law gives the magnetic field $d\vec{B}$ due to a small current element $Id\vec{\ell}$:
where $\mu_0 = 4\pi \times 10^{-7}$ T m A⁻¹ is the permeability of free space. Important results derived using Biot–Savart:
| Configuration | Magnetic Field | Location |
|---|---|---|
| Long straight wire carrying I | $B = \dfrac{\mu_0 I}{2\pi r}$ | At perpendicular distance r |
| Circular loop of radius R, current I | $B = \dfrac{\mu_0 I}{2R}$ | At centre of loop |
| Solenoid (n turns/m, current I) | $B = \mu_0 nI$ | Inside (uniform field) |
| Toroid (N turns, current I, radius r) | $B = \dfrac{\mu_0 NI}{2\pi r}$ | Inside the toroid |
Ampere's Circuital Law
Ampere's Law is the magnetic analogue of Gauss's Law. For a closed path (Amperian loop) enclosing total current $I_{enc}$:
This law is most useful for highly symmetric current distributions (infinite straight wire, solenoid, toroid). The force per unit length between two parallel wires carrying currents $I_1$ and $I_2$ separated by distance $d$ is:
Currents in the same direction attract; currents in opposite directions repel. This is the basis for the SI definition of the Ampere.
Galvanometer, Ammeter, and Voltmeter
A galvanometer detects small currents. To convert it into an ammeter (low resistance, in series with circuit), a shunt $S$ is connected in parallel: $S = \frac{I_g G}{I - I_g}$. To convert it into a voltmeter (high resistance, in parallel with circuit), a high resistance $R$ is connected in series: $R = \frac{V}{I_g} - G$, where $G$ is the galvanometer resistance and $I_g$ is its full-scale deflection current.
Chapter 5 – Magnetism and Matter
Materials respond to magnetic fields differently. The magnetisation $M$ is the magnetic dipole moment per unit volume. The magnetic susceptibility $\chi_m = M/H$ classifies materials:
- Diamagnetic ($\chi_m$ small and negative): Weakly repelled by magnets; examples: bismuth, copper, water. No permanent dipoles.
- Paramagnetic ($\chi_m$ small and positive): Weakly attracted; examples: aluminium, platinum. Random atomic dipoles align slightly with external field. Obeys Curie's Law: $\chi_m = C/T$.
- Ferromagnetic ($\chi_m$ large and positive): Strongly attracted; examples: iron, cobalt, nickel. Permanent domains; shows hysteresis.
Unit IV: Electromagnetic Induction and Alternating Current
In 1831, Michael Faraday discovered that a changing magnetic field creates an electric field—electromagnetic induction. This single discovery is responsible for every electric generator, transformer, and inductor on Earth. Chapter 6 (EMI) and Chapter 7 (AC) together form the engine of modern power systems.
Chapter 6 – Electromagnetic Induction
Magnetic Flux through a surface is $\Phi = \int \vec{B} \cdot d\vec{A} = BA\cos\theta$, measured in Weber (Wb). Faraday's First Law: An EMF is induced in a coil whenever the magnetic flux through it changes. Faraday's Second Law: The magnitude of the induced EMF equals the rate of change of flux:
The negative sign embodies Lenz's Law: the induced current always opposes the change that caused it (a consequence of energy conservation). For a rectangular coil of $N$ turns, area $A$, rotating at angular velocity $\omega$ in field $B$: $\varepsilon = NBA\omega\sin\omega t = \varepsilon_0 \sin\omega t$.
Self-Inductance ($L$): A coil opposes changes in its own current. $\varepsilon = -L\frac{dI}{dt}$. For a solenoid: $L = \mu_0 n^2 V$ (V = volume). Mutual Inductance ($M$): Two coils coupled so that changing current in coil 1 induces EMF in coil 2: $\varepsilon_2 = -M\frac{dI_1}{dt}$. The energy stored in an inductor is:
Chapter 7 – Alternating Current
AC voltage and current vary sinusoidally with time. The RMS (root mean square) values are what we use for power calculations: $V_{rms} = V_0/\sqrt{2}$ and $I_{rms} = I_0/\sqrt{2}$. The average power in an AC circuit is $P = V_{rms} I_{rms} \cos\phi$, where $\phi$ is the phase angle between voltage and current and $\cos\phi$ is the power factor.
| Circuit Element | Reactance/Impedance | Phase (V leads I by) |
|---|---|---|
| Pure Resistor (R) | Z = R | 0° (in phase) |
| Pure Inductor (L) | $X_L = \omega L = 2\pi f L$ | +90° |
| Pure Capacitor (C) | $X_C = \dfrac{1}{\omega C}$ | −90° |
| Series RLC | $Z = \sqrt{R^2 + (X_L - X_C)^2}$ | $\tan\phi = \dfrac{X_L - X_C}{R}$ |
Resonance in a series RLC circuit occurs when $X_L = X_C$, i.e., $\omega_0 = 1/\sqrt{LC}$. At resonance, impedance is minimum ($Z = R$), current is maximum, and power factor equals 1. The quality factor $Q = \omega_0 L / R$ measures sharpness of resonance.

A transformer uses mutual induction to change voltage levels. For an ideal transformer with $N_p$ primary turns and $N_s$ secondary turns: $\frac{V_s}{V_p} = \frac{N_s}{N_p} = \frac{I_p}{I_s}$. Step-up: $N_s > N_p$ (increases voltage, decreases current); step-down: $N_s < N_p$. Real transformers have losses from eddy currents, hysteresis, and resistance heating.
Unit V: Electromagnetic Waves
In 1865, James Clerk Maxwell unified electricity, magnetism, and optics by predicting the existence of electromagnetic waves—self-sustaining oscillating electric and magnetic fields that travel through space at the speed of light. This was one of the greatest intellectual achievements in the history of science.
Maxwell's Displacement Current
Ampere's Law worked perfectly for steady currents. But Maxwell noticed an inconsistency when applied to a charging capacitor: current flows in the wires but not between the plates, yet a magnetic field clearly exists there. Maxwell introduced the displacement current $I_d = \varepsilon_0 \frac{d\Phi_E}{dt}$ to complete the picture. This modifies Ampere's Law to:
Properties of EM Waves
- EM waves are transverse: $\vec{E}$ and $\vec{B}$ are perpendicular to each other and to the direction of propagation.
- They travel in vacuum at speed $c = \frac{1}{\sqrt{\mu_0 \varepsilon_0}} = 3 \times 10^8$ m/s.
- At every point, $E_0 / B_0 = c$.
- They carry both energy and momentum but do not require a medium (they travel through vacuum).
- Intensity (average power per unit area): $I = \frac{1}{2} c \varepsilon_0 E_0^2$.
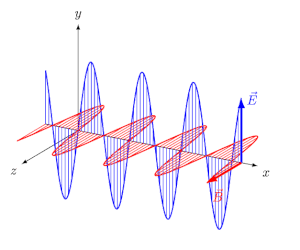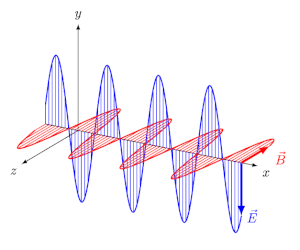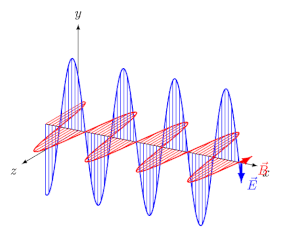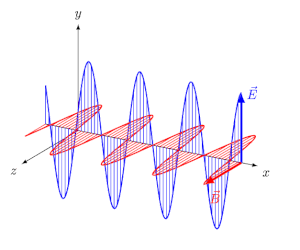
The Electromagnetic Spectrum
| Type | Wavelength Range | Source / Use |
|---|---|---|
| Radio Waves | > 0.1 m | Oscillating circuits; broadcasting, radar |
| Microwaves | 1 mm – 0.1 m | Klystron, magnetron; microwave ovens, satellite communication |
| Infrared | 700 nm – 1 mm | Hot bodies; remote controls, thermal imaging |
| Visible Light | 400 – 700 nm | Excited atoms; human vision |
| Ultraviolet | 1 – 400 nm | Sun, UV lamps; sterilisation, photography |
| X-rays | 0.01 – 10 nm | X-ray tubes, synchrotrons; medical imaging |
| Gamma Rays | < 0.01 nm | Radioactive nuclei; cancer treatment |
Unit VI: Optics — Ray Optics and Wave Optics
Optics (Chapters 9 & 10) is the study of light and its interactions with matter. It is split into ray (geometric) optics—which treats light as rays obeying simple reflection and refraction laws—and wave optics—which explains interference, diffraction, and polarisation using the wave nature of light. This unit carries the highest combined marks (18) and contains many derivation-based questions.
Chapter 9 – Ray Optics and Optical Instruments
Reflection at a curved mirror follows the mirror formula:
where $u$ = object distance, $v$ = image distance, $f$ = focal length, $R$ = radius of curvature. Linear magnification: $m = -v/u$. Sign convention: distances measured from the pole of the mirror; distances in the direction of incident light are positive.
Refraction occurs when light passes from one medium to another. Snell's Law:
Total Internal Reflection (TIR) occurs when light travels from denser to rarer medium and the angle of incidence exceeds the critical angle $\theta_c = \sin^{-1}(n_2/n_1)$. TIR is exploited in optical fibres, diamonds, and binoculars.
For a thin lens, the lens maker's equation and lens formula are:
Power of a lens: $P = 1/f$ (f in metres), measured in Dioptres (D). For lenses in contact: $P = P_1 + P_2$.
| Optical Instrument | Key Formula | Magnification (normal adjustment) |
|---|---|---|
| Simple Microscope | Uses convex lens as magnifier | $m = 1 + D/f$ (D = 25 cm) |
| Compound Microscope | Objective + eyepiece in series | $m = m_o \times m_e = \frac{L}{f_o} \times \frac{D}{f_e}$ |
| Astronomical Telescope | Refracting; objective large $f_o$ | $m = -f_o / f_e$ |
| Reflecting Telescope | Concave mirror as objective | No chromatic aberration |
Chapter 10 – Wave Optics
Wave optics describes phenomena that ray optics cannot: interference, diffraction, and polarisation. Huygen's Principle states that every point on a wavefront acts as a secondary source of spherical wavelets; the new wavefront is the tangent envelope of these wavelets.
Young's Double Slit Experiment (YDSE): Two coherent sources separated by distance $d$, placed at distance $D$ from screen, produce bright fringes (constructive interference) and dark fringes (destructive interference). Fringe width:
Position of $n$th bright fringe: $y_n = n\frac{\lambda D}{d}$. Position of $n$th dark fringe: $y_n = (2n-1)\frac{\lambda D}{2d}$. The intensity distribution is $I = 4I_0 \cos^2(\delta/2)$ where $\delta$ is the phase difference.

Single Slit Diffraction: A slit of width $a$ produces a central maximum of width $2\lambda D/a$ and minima at $a\sin\theta = m\lambda$ ($m = \pm 1, \pm 2, \ldots$). The central maximum is twice as wide as the secondary maxima.
Polarisation: Light is a transverse wave. Natural (unpolarised) light has $\vec{E}$ oscillating in all directions perpendicular to propagation. A Polaroid transmits only one plane of oscillation. Malus's Law: $I = I_0 \cos^2\theta$, where $\theta$ is the angle between the polariser and analyser transmission axes. Brewster's Law: At the polarising angle $\theta_B$, reflected light is completely polarised: $\tan\theta_B = n$.
Unit VII: Dual Nature of Radiation and Matter
By the late 19th century, experiments showed that light couldn't be purely a wave (photoelectric effect) and that matter couldn't be purely particle-like (electron diffraction). The resolution was profound: light and matter both have wave-particle duality. This unit (Chapter 11, 12 marks) introduced quantum mechanics to the world.
The Photoelectric Effect
When light of sufficient frequency shines on a metal surface, electrons are emitted. Einstein's explanation (1905, Nobel Prize 1921): light consists of photons, each of energy $E = h\nu$ (where $h = 6.626 \times 10^{-34}$ J s is Planck's constant). An electron absorbs one photon; if $h\nu$ exceeds the work function $\phi_0$ (minimum energy to free the electron), the electron is emitted with maximum kinetic energy:
where $V_0$ is the stopping potential (minimum retarding voltage to stop all emitted electrons). Key experimental observations: (1) Emission starts only above a threshold frequency $\nu_0 = \phi_0/h$, regardless of intensity. (2) $K_{max}$ depends on frequency, not intensity. (3) Emission is instantaneous ($< 10^{-9}$ s). All three facts are inconsistent with classical wave theory but perfectly explained by the photon model.
de Broglie Hypothesis: Matter Waves
Louis de Broglie (1924) proposed that matter, like light, has a wave character. The de Broglie wavelength of a particle with momentum $p = mv$ is:
For an electron accelerated through potential difference $V$: $\lambda = \frac{h}{\sqrt{2meV}} = \frac{1.227}{\sqrt{V}}$ nm. The Davisson–Germer experiment (1927) confirmed de Broglie's hypothesis by showing that electrons diffract from a crystal just like X-rays.
| Concept | Classical Picture | Quantum (Photon/Wave) Picture |
|---|---|---|
| Nature of light | Continuous wave; energy spread uniformly | Discrete photons; $E = h\nu$ |
| Energy depends on | Amplitude (intensity) | Frequency ($\nu$) |
| Threshold frequency | Should not exist | Exists: $\nu_0 = \phi_0 / h$ |
| Time delay | Long delay expected | Instantaneous emission |
Unit VIII: Atoms and Nuclei
Chapters 12 and 13 take us inside the atom—first to the electron orbits, then into the nucleus itself. These chapters connect directly to modern technology: nuclear power, radiocarbon dating, PET scans, and radiation therapy all rest on the physics explored here.
Chapter 12 – Atoms: The Bohr Model
Rutherford's 1911 scattering experiment established the nuclear model of the atom: a tiny, massive, positively charged nucleus surrounded by electrons. Niels Bohr (1913) added quantum postulates to explain the discrete spectrum of hydrogen. Bohr's Postulates: (1) Electrons occupy only certain allowed orbits where the angular momentum is quantised: $mvr = \frac{nh}{2\pi} = n\hbar$. (2) Electrons in allowed orbits do not radiate. (3) Radiation is emitted or absorbed when an electron transitions between orbits.
For hydrogen (atomic number $Z=1$), the key results are:
The ground state energy is $E_1 = -13.6$ eV (negative because the electron is bound). When an electron falls from level $n_2$ to $n_1$, the emitted photon has frequency:
where $R_H = 1.097 \times 10^7$ m⁻¹ is the Rydberg constant. The spectral series are: Lyman ($n_1=1$, UV), Balmer ($n_1=2$, visible), Paschen ($n_1=3$, IR), Brackett ($n_1=4$, IR), Pfund ($n_1=5$, IR).

Chapter 13 – Nuclei: Radioactivity and Nuclear Reactions
The nucleus consists of protons and neutrons (collectively nucleons) held together by the strong nuclear force—far stronger than electromagnetic repulsion but very short-ranged (~1–3 fm). Nuclear size follows $R = R_0 A^{1/3}$ with $R_0 \approx 1.2$ fm, $A$ = mass number. The nuclear density (~$2.3 \times 10^{17}$ kg/m³) is constant for all nuclei.
Mass Defect and Binding Energy: The actual mass of a nucleus is less than the sum of its constituent nucleon masses. This missing mass, the mass defect $\Delta m$, is converted to binding energy $E_B = \Delta m \cdot c^2$ (using Einstein's $E = mc^2$). The binding energy per nucleon ($E_B/A$) peaks around iron-56 (~8.8 MeV/nucleon), explaining why both fusion (light nuclei fusing) and fission (heavy nuclei splitting) release energy.
Radioactivity: Unstable nuclei decay spontaneously. The three main types are:
- Alpha (α) decay: Emission of $^4_2$He nucleus. A decreases by 4, Z decreases by 2. Least penetrating; stopped by paper.
- Beta (β⁻) decay: Emission of electron and antineutrino; a neutron converts to a proton. Z increases by 1, A unchanged. Stopped by a few mm of aluminium.
- Gamma (γ) decay: Emission of high-energy photon from excited nucleus. A and Z unchanged. Most penetrating; requires thick lead or concrete shielding.
Radioactive decay follows an exponential law:
where $\lambda$ is the decay constant and $T_{1/2}$ is the half-life (time for half the nuclei to decay). Activity $A = \lambda N$, measured in Becquerel (Bq = 1 decay/s) or Curie ($1$ Ci $= 3.7 \times 10^{10}$ Bq).
Unit IX: Semiconductor Devices — Diodes, Transistors and Logic Gates
Every smartphone, computer, and digital gadget runs on semiconductor physics. Chapter 14 (7 marks) introduces the quantum-mechanical basis of how silicon and other semiconductors work, and how tiny junctions between differently-doped materials can control enormous currents—forming the transistors that power the modern world.
Energy Bands and Semiconductor Types
In a solid, atomic energy levels broaden into bands. The valence band is the highest filled band; the conduction band is above it. The band gap $E_g$ separates them. For conductors: $E_g = 0$ (bands overlap). For semiconductors: $E_g \approx 0.1 – 3$ eV (e.g., Si: 1.1 eV, Ge: 0.7 eV). For insulators: $E_g > 3$ eV.
An intrinsic semiconductor (pure crystal) has equal numbers of electrons (n) in the conduction band and holes (p) in the valence band: $n = p = n_i$, where $n_i$ increases sharply with temperature. Doping adds impurities: adding pentavalent atoms (P, As) creates n-type (extra electrons); adding trivalent atoms (B, In) creates p-type (extra holes). In both cases, $np = n_i^2$ at equilibrium.
The p-n Junction and Diode
When p-type and n-type materials are joined, electrons diffuse to the p-side and holes diffuse to the n-side, leaving behind charged ions. This creates a depletion region with a built-in electric field (contact potential $V_0 \approx 0.6$–$0.7$ V for Si) that opposes further diffusion. In forward bias (p connected to +), the barrier decreases and current flows easily. In reverse bias (p connected to −), the barrier increases and only a tiny reverse saturation current flows until breakdown.

Rectification: A diode converts AC to DC. In a half-wave rectifier, only one half-cycle passes. In a full-wave rectifier (bridge circuit with 4 diodes), both halves are rectified. A Zener diode is specially doped to undergo controlled breakdown at a precise Zener voltage $V_Z$; it is used in voltage regulators to maintain constant output voltage.
Transistors and Logic Gates
A bipolar junction transistor (BJT) has three doped regions: Emitter (heavily doped), Base (very thin, lightly doped), and Collector. In the common-emitter (CE) configuration, a small base current $I_B$ controls a much larger collector current $I_C$. The current gain $\beta = I_C / I_B$ is typically 50–300. The transistor acts as an amplifier (in active region) or switch (in saturation/cut-off). Voltage gain of CE amplifier: $A_v = -\beta \frac{R_C}{R_{in}}$.
| Logic Gate | Boolean Expression | Output Rule |
|---|---|---|
| NOT | $Y = \bar{A}$ | Output is complement of input |
| AND | $Y = A \cdot B$ | Output 1 only if both inputs are 1 |
| OR | $Y = A + B$ | Output 1 if at least one input is 1 |
| NAND | $Y = \overline{A \cdot B}$ | Output 0 only if both inputs are 1 |
| NOR | $Y = \overline{A + B}$ | Output 1 only if both inputs are 0 |
Exam Strategy, High-Weightage Derivations & Formula Quick-Reference
Class 12 Physics board exams consistently reward students who (a) understand concepts deeply enough to apply them to new situations, (b) know which derivations are asked repeatedly, and (c) present solutions clearly with diagrams and proper units. Use this final section as your revision checklist.
Must-Know Derivations (5-mark questions)
- Electric field due to a dipole on axial and equatorial positions
- Gauss's Law application: field due to uniformly charged spherical shell
- Drift velocity and resistance: deriving $R = \rho L/A$ from microscopic view
- Magnetic field on axis of circular loop using Biot–Savart Law
- Faraday's Law: EMF of a rotating rectangular coil in a uniform magnetic field
- Lens Maker's Equation using refraction at two spherical surfaces
- Young's Double Slit: fringe width derivation
- Bohr's model: derivation of $r_n$ and $E_n$ for hydrogen
- de Broglie wavelength for an accelerated electron
- Transistor as amplifier in CE configuration with voltage gain derivation
Ultimate Formula Quick-Reference
| Chapter | Formula | Symbol Guide |
|---|---|---|
| Coulomb's Law | $F = kq_1q_2/r^2$ | k = 9×10⁹ N m² C⁻² |
| Electric Field (point charge) | $E = kq/r^2$ | r = distance from charge |
| Capacitance (parallel plate) | $C = K\varepsilon_0 A/d$ | K = dielectric constant |
| Ohm's Law | $V = IR$ | R = resistance in Ω |
| Magnetic Force on current | $F = BIL\sin\theta$ | L = length of conductor |
| Solenoid field | $B = \mu_0 nI$ | n = turns per metre |
| Induced EMF | $\varepsilon = -N\,d\Phi/dt$ | N = number of turns |
| RLC Impedance | $Z = \sqrt{R^2+(X_L-X_C)^2}$ | $X_L=\omega L$, $X_C=1/\omega C$ |
| Lens Formula | $1/v - 1/u = 1/f$ | Sign convention: incident light → +ve |
| YDSE Fringe Width | $\beta = \lambda D/d$ | D = screen distance, d = slit separation |
| Photoelectric Effect | $K_{max} = h\nu - \phi_0$ | h = 6.626×10⁻³⁴ J s |
| de Broglie Wavelength | $\lambda = h/mv$ | p = mv = momentum |
| Bohr Energy Level | $E_n = -13.6/n^2$ eV | n = principal quantum number |
| Radioactive Decay | $N = N_0 e^{-\lambda t}$ | $T_{1/2} = 0.693/\lambda$ |
| Transistor Current Gain | $\beta = I_C / I_B$ | Also: $\alpha = I_C/I_E$; $\beta = \alpha/(1-\alpha)$ |
Time-Saving Exam Techniques
- Read questions carefully: Many students lose marks by answering what they assumed was asked, not what was actually asked. Underline key words.
- Diagrams earn marks: In optics and magnetism, a clearly labelled diagram often accounts for 1–2 marks of a 5-mark question.
- Units in every answer: Writing the unit of your final answer is mandatory. Dimensionless answers should still state 'dimensionless' if that's correct.
- SI units throughout: Convert all given quantities to SI before substituting into formulas to avoid factor-of-1000 errors.
- Start with high-weightage questions you know: This secures marks early and reduces anxiety. Skip and return to uncertain questions.
- State the law/principle before deriving: In derivation questions, always state the law you're applying (e.g., 'Applying Gauss's Law to the Gaussian surface…') before the mathematical steps.